Sterilization is fundamental to maintaining patient safety in every clinical setting, greatly reducing the risk of infection and helping deliver positive results. While single-use products are common, many essential and high-value devices are built for reuse and must undergo robust cleaning and sterilization between each use. Explore eight reusable medical devices compatible with sterilization and why validated reprocessing procedures matter.
Endoscopes for Minimally Invasive Procedures
Healthcare providers use endoscopes to examine internal organs during minimally invasive procedures. With features like long internal channels, delicate lenses, and electronic components, they present significant challenges during cleaning and sterilization. Consistent, thorough reprocessing of endoscopes is essential to reduce infection risks.
Manufacturers are required to create detailed, validated instructions for use (IFUs) that healthcare staff can reliably follow. These IFUs cover every step, from manual cleaning techniques to the specific type of sterilization suitable for the materials and construction of the device. Validation ensures that the procedures detailed in the IFU consistently remove contaminants and adequately sterilize the instrument.
Surgical Trays for Instrument Organization
Healthcare professionals use surgical trays for grouping, transporting, and sterilizing surgical instruments during procedures. Built from stainless steel or robust polymers, trays are engineered to withstand repeated steam sterilization and other common methods used in hospital settings. When properly wrapped with the appropriate sterilization wrapper, their open designs allow the sterilizing agent to reach all surfaces.
Staff need to follow best practices for loading trays to make sure the sterilant can contact every instrument. Overpacking can lead to incomplete sterilization, putting patient safety at risk. During validation, trays are tested using a heavy weight sterilization wrap (worse case) to confirm that their design does not hinder the sterilization of the tools they carry.
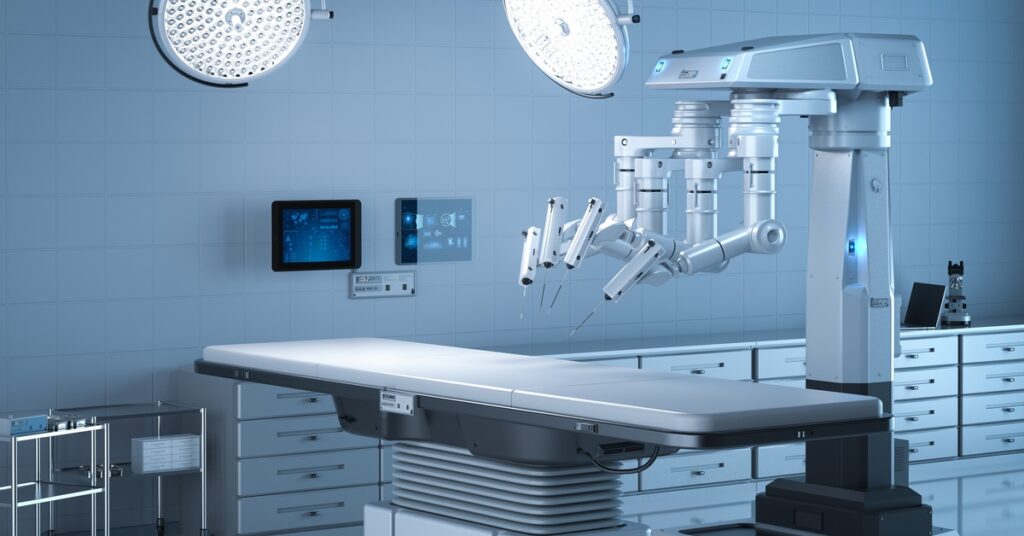
Robotic Surgery Instruments
Robotic instruments are at the leading edge of surgical technology, enabling greater precision and dexterity during operations. These advanced, high-value tools are intended for repeated reuse and require effective, validated reprocessing to ensure safe performance. Their sophisticated construction often incorporates moving joints and electronic elements, all of which add complexity to the cleaning and sterilization process.
For robotic instruments, manufacturers must use rigorous, evidence-based methods to design and validate cleaning and sterilization guidelines. Medical device validation testing is the structured process used to establish that a manufacturer’s recommended cleaning, disinfection, and sterilization steps are both effective and reproducible, thus supporting safe device reuse. At Highpower Labs, we document the results of these validation processes, providing our clients with the data needed to support regulatory submissions. This information can then be incorporated into master device files to demonstrate compliance and ensure safe reprocessing practices.
Challenges in Sterilizing Robotic Tools
Robotic devices such as intricate endoscopic instruments and graspers pose unique cleaning challenges. Their construction can allow debris to lodge in joints or crevices, which may require the use of specialized cleaning brushes, enzymatic cleaners, or ultrasonic baths before sterilization. Only materials proven compatible with hospital sterilization—such as steam or vaporized hydrogen peroxide—are selected, ensuring repeated use won’t degrade device performance.
Orthopedic Implants and Tools
Orthopedic procedures frequently make use of reusable tools and accessories like bone saws, drills, and surgical guides. As these instruments come into direct contact with bone and tissue, preventing any contamination is critical to patient safety.
Cleaning protocols must demonstrate the ability to remove all biological debris before sterilization is carried out. Manufacturers usually supply practical guides for assembling, cleaning, and inspecting these tools.
Typical standards for orthopedic devices include:
- Endurance of repeated steam autoclave cycles.
- Protection against corrosion from various cleaning agents.
- Clear procedures for both manual and automated cleaning.
- Guidelines for thorough inspection after cleaning.
- Tips for storage and packaging to maintain sterility until use.
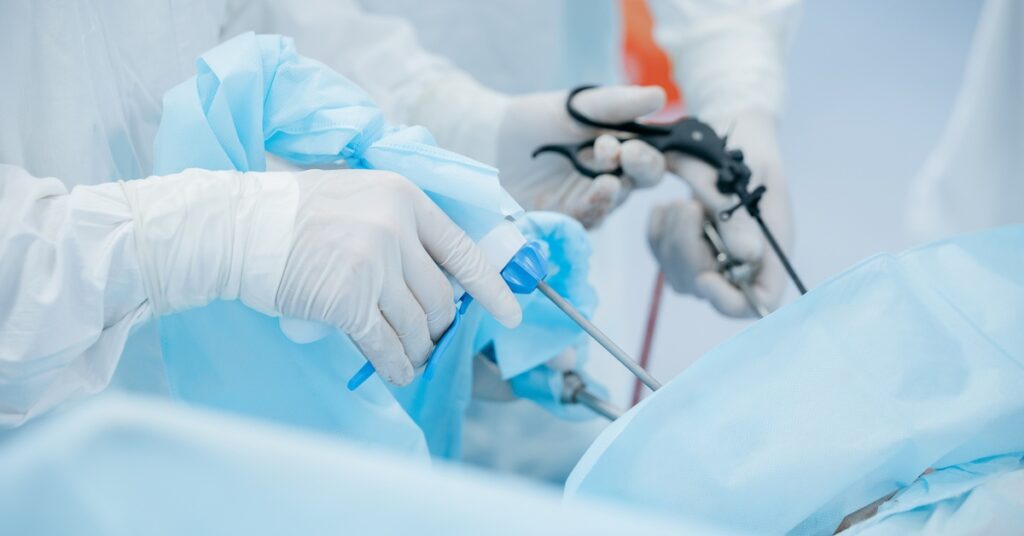
Laparoscopic Instruments
Laparoscopic tools—like graspers, scissors, and dissectors—are critical for minimally invasive abdominal surgeries. Their unique design, which often features slim shafts and complex working ends, makes thorough cleaning and sterilization more demanding than for conventional instruments. The entire internal and external surface must be sterilized to prevent infections.
Manufacturers must validate that their specified cleaning and sterilization instructions are realistic and reliable for hospital staff to follow. If cleaning protocols can’t reliably reach every surface or lumen, contaminants may remain, heightening cross-contamination risks. Consequently, regulatory agencies place considerable emphasis on validated IFUs and consistent reprocessing results.
Dental Handpieces
Dental handpieces—such as high-speed drills and polishers—are among the most consistently used reusable devices in dental care. Each comes into close contact with blood, tissue, and fluids, making consistent sterilization between patients mandatory. Their intricate turbines, internal tubing, and gear systems make cleaning and sanitization more complex.
Manufacturers address these challenges by developing and validating step-by-step cleaning, flushing, and sterilization instructions. Following these not only protects patients but also helps the equipment operate effectively over time. Adhering to recognized standards supports both compliance and infection prevention efforts in the dental field.
Reusable Scalpels and Forceps
Nearly all surgical teams rely on reusable scalpels and forceps, medical devices that are compatible with sterilization. They’re typically manufactured from high-grade stainless steel, ensuring sharpness and resilience through repeated steam cycles. Even with the availability of disposable alternatives, the reusable versions offer significant cost and sustainability advantages.
Despite their simpler shapes, strict processing procedures are essential to successful sterilization. All visible debris must be removed before items are packaged and sterilized. Validating cleaning and sterilization steps is just as vital for these tools as it is for more complicated devices.
Focus factors for reprocessing include:
- Choosing suitable, laboratory-tested detergents.
- Applying manual or automated cleaning methods as specified.
- Visually inspecting for any residue or soil.
- Packaging instruments in a way that allows the efficient flow of sterilant.
- Using validated sterilization cycles tailored for these products.
Retractors Used in Surgery
Retractors, essential for holding tissue or organs aside during surgery, are used across all surgical specialties. These come in a variety of forms, from compact hand-held hooks to large self-retaining frames, all built for repeated, reliable use. Designed with robust materials, retractors must undergo thorough reprocessing due to their deep tissue contact.
Ensuring these instruments are sterile is vital for preventing infection, as any biological residue presents a higher risk. Validation testing for retractors covers both the effectiveness of cleaning procedures and the sterilization steps needed to reach a satisfactory sterility assurance level. Devices with hinges or multiple components often require specialized cleaning techniques to ensure no surfaces are missed.
Bringing a reusable medical device into routine clinical use involves layered safety requirements, detailed regulatory review, and technical validation. Validation confirms that the sterilization process is both effective and feasible for routine use by healthcare professionals. Trust Highpower Labs to provide the detailed validation testing your reusable medical devices need for safe and compliant clinical use. Through comprehensive validation studies, manufacturers and healthcare providers support the highest levels of infection control.
